Buffer solution 1 M pH 8.5 (volatile), Triethylammonium hydrogen carbonate buffer
triethylammonium hydrogen carbonate buf-fer 1.0 M in water
CAS: 15715-58-9
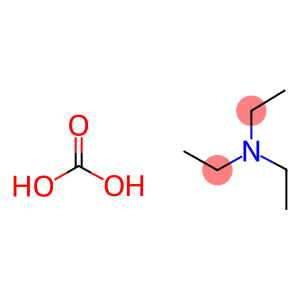
Molecular Formula: C7H17NO3
Buffer solution 1 M pH 8.5 (volatile), Triethylammonium hydrogen carbonate buffer - Names and Identifiers
Buffer solution 1 M pH 8.5 (volatile), Triethylammonium hydrogen carbonate buffer - Physico-chemical Properties
| Molecular Formula | C7H17NO3 |
| Molar Mass | 163.21 |
| Density | 1.02g/mLat 25°C(lit.) |
| Melting Point | 17 °C (decomp) |
| Boling Point | 314.8°C at 760 mmHg |
| Flash Point | 144.2°C |
| Vapor Presure | 0.000456mmHg at 25°C |
| Appearance | Liquid |
| Maximum wavelength(λmax) | ['λ: 260 nm Amax: ≤0.05', , 'λ: 280 nm Amax: ≤0.05'] |
| BRN | 3624751 |
| PH | 8.4-8.6 (25℃) |
| Storage Condition | 2-8°C |
| Refractive Index | n20/D 1.355 |
Buffer solution 1 M pH 8.5 (volatile), Triethylammonium hydrogen carbonate buffer - Risk and Safety
| Hazard Symbols | C - Corrosive |
| Risk Codes | R34 - Causes burns R42 - May cause sensitization by inhalation |
| Safety Description | S23 - Do not breathe vapour. S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. S45 - In case of accident or if you feel unwell, seek medical advice immediately (show the label whenever possible.) |
| UN IDs | UN 3265 8/PG 3 |
| WGK Germany | 3 |
| HS Code | 29239000 |
Buffer solution 1 M pH 8.5 (volatile), Triethylammonium hydrogen carbonate buffer - Introduction
buf-fer 1.0 M in water, also known as TEABHCO3, is an organic compound. The following is a description of its nature, use, preparation and safety information:
Nature:
1. Appearance: triethylammonium hydrogen carbonate buf-fer 1.0 M in water is white crystalline powder.
2. Solubility: it can be dissolved in water, moderate solubility.
3. Acidity and alkalinity: triethylammonium hydrogen carbonate buf-fer 1.0 M in water is a weakly acidic compound that can react with alkali to generate salt and carbon dioxide.
Use:
1.triethylammonium hydrogen carbonate buf-fer 1.0 M in water is commonly used as a catalyst for organic synthesis and shows good catalytic activity in organic reactions.
2. It can also be used as a climate control agent for electrolytes in thermocouple calculation instruments.
3. in the synthesis of some drugs, triethylammonium hydrogen carbonate buf-fer 1.0 M in water is a commonly used alkaline agent.
Method:
The preparation of triethylammonium hydrogen carbonate buf-fer 1.0 M in water is usually obtained by the reaction of diethyl carbonate and ammonium hydroxide. The specific steps include: dissolving diethyl carbonate in a suitable solvent, then slowly adding ammonium hydroxide solution Dropwise, and reacting at a suitable temperature and stirring for a certain time. Finally, filtration, washing and drying gave the triethylammonium hydrogen carbonate buf-fer 1.0 M in water product.
Safety Information:
1.triethylammonium hydrogen carbonate buf-fer 1.0 M in water is relatively stable at room temperature, but it may decompose at high temperature and fire source, releasing toxic gas.
2. Use should avoid contact with strong oxidants and strong acids to avoid dangerous reactions.
3. Avoid inhalation or direct contact with skin and eyes, if there is contact, should be timely cleaning.
4. During storage, keep the triethylammonium hydrogen carbonate buf-fer 1.0 M in water sealed in a cool and dry place, away from fire and combustibles.
Please note that the above information is for reference only, and the specific operation and use should be carried out according to actual needs and relevant safety regulations.
Nature:
1. Appearance: triethylammonium hydrogen carbonate buf-fer 1.0 M in water is white crystalline powder.
2. Solubility: it can be dissolved in water, moderate solubility.
3. Acidity and alkalinity: triethylammonium hydrogen carbonate buf-fer 1.0 M in water is a weakly acidic compound that can react with alkali to generate salt and carbon dioxide.
Use:
1.triethylammonium hydrogen carbonate buf-fer 1.0 M in water is commonly used as a catalyst for organic synthesis and shows good catalytic activity in organic reactions.
2. It can also be used as a climate control agent for electrolytes in thermocouple calculation instruments.
3. in the synthesis of some drugs, triethylammonium hydrogen carbonate buf-fer 1.0 M in water is a commonly used alkaline agent.
Method:
The preparation of triethylammonium hydrogen carbonate buf-fer 1.0 M in water is usually obtained by the reaction of diethyl carbonate and ammonium hydroxide. The specific steps include: dissolving diethyl carbonate in a suitable solvent, then slowly adding ammonium hydroxide solution Dropwise, and reacting at a suitable temperature and stirring for a certain time. Finally, filtration, washing and drying gave the triethylammonium hydrogen carbonate buf-fer 1.0 M in water product.
Safety Information:
1.triethylammonium hydrogen carbonate buf-fer 1.0 M in water is relatively stable at room temperature, but it may decompose at high temperature and fire source, releasing toxic gas.
2. Use should avoid contact with strong oxidants and strong acids to avoid dangerous reactions.
3. Avoid inhalation or direct contact with skin and eyes, if there is contact, should be timely cleaning.
4. During storage, keep the triethylammonium hydrogen carbonate buf-fer 1.0 M in water sealed in a cool and dry place, away from fire and combustibles.
Please note that the above information is for reference only, and the specific operation and use should be carried out according to actual needs and relevant safety regulations.
Last Update:2024-04-09 21:00:56
Supplier List
Spot supply
Product Name: TRIETHYLAMMONIUM FORMATE Visit Supplier Webpage Request for quotationCAS: 15715-58-9
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: TRIETHYLAMMONIUM FORMATE Request for quotation
CAS: 15715-58-9
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 15715-58-9
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Spot supply
Product Name: TRIETHYLAMMONIUM FORMATE Visit Supplier Webpage Request for quotationCAS: 15715-58-9
Tel:
Email: qianyanbiochem@gmail.com
Mobile: 13247110337
QQ: 2972965813
Product List: View Catalog
Product Name: TRIETHYLAMMONIUM FORMATE Request for quotation
CAS: 15715-58-9
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 15715-58-9
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
View History

